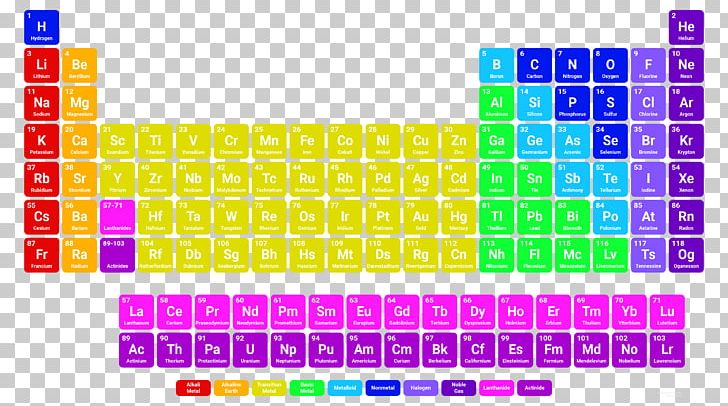
The Non-Metal topic has been briefly discussed, right from Non-Metal definition to Non-Metal examples. Sulphur is utilised to make Sulphuric acid. Chlorine has the best use for contaminated umpire water. Nitrogen is used to make fertilisers, nitric acid, and ammonia. The Non-Metals are:Īs discussed above, Non-Metal can be used to make various substances like hydrogen, which helps prepare fuel used in rockets. There are a total of 20 Non-Metal examples which are given in the periodic table. For example, carbon dioxide, when reacted with water given carbonic acid. When a Non-Metal reacts with water, it gives rise to acid.When a Non-Metal reacts in the air, like if carbon reacts with oxygen, it gives rise to carbon monoxide, carbon dioxide, etc.Non-Metal goes through an oxidation reaction in the presence of oxygen to form oxides(neutral or acidic).The metal and Non-Metal give rise to ionic bonds where the metal gets oxidised and the Non-Metal gets reduced.A Non-Metal like chlorine reacts with a metal, sodium, to form sodium chloride, a salt.Non-Metal doesn’t react with water while in the air, especially with oxygen.It can occur as diatomic, monoatomic, and polyatomic molecules.It exists as solid and gases under normal pressure or room temperature.It generally does not lose its electrons quickly because of their electronegativity.Graphite is an exception here as it is one of the best conductors of heat and electricity, even being a Non-Metal. It is a poor conductor of electricity and heat.When moving from left to right of the table, the Non-Metal starts having more Non-Metallic properties as it gains more electrons in its valence shell.It is non-sonorous, so they don’t create ringing sounds when beaten or hitting any surface.It doesn’t have any definite shape or size and can be easily broken.This is why Non-Metals are electronegative, which means the negative ions formed during the formation of a bond. Non-Metal in their outer shell contains more than four electrons.Let’s discuss the physical and chemical properties of Non-Metal. Along with that, Non-Metal is non-lustrous. They are generally poor conductors of heat and electricity, but there are certain exceptions. Non-Metal is a substance that shares or gains electrons which forms negative ions during a chemical reaction. In this class, Non-Metal exists in either of the forms, which is monatomic anions and a part of polyatomic anions. It forms ions by adding or extracting electrons from its atom. 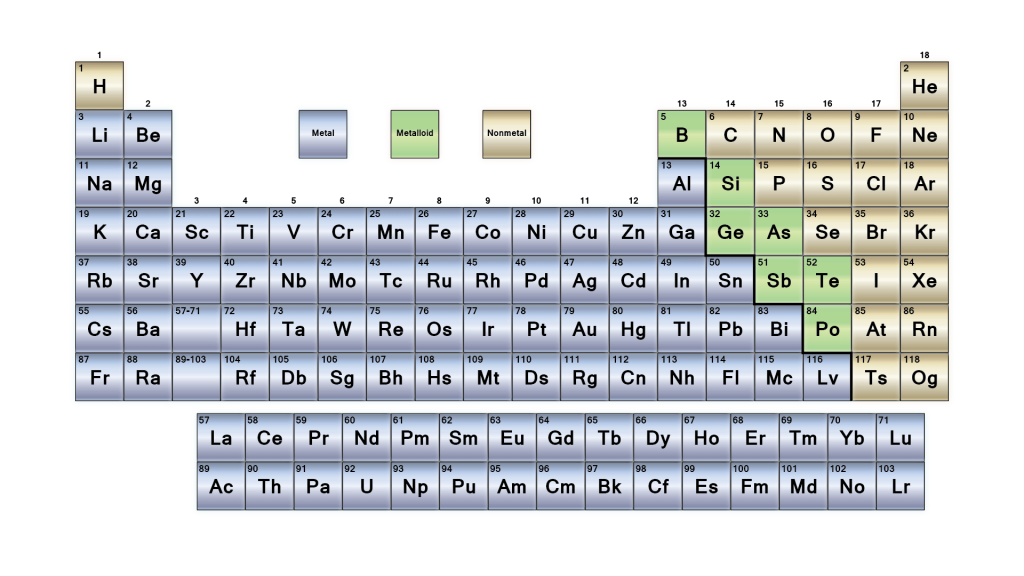 
In the other class, ionic materials are Non-Metal that involve minor and significant atoms in them. The covalent materials include atoms smaller in size with a high rate of negative ions, along with negative oxidation in their compounds. Non-Metal can be classified into two classes, one is covalent materials and the other one is ionic materials. They react precisely opposite to the metals. The Non-Metal definition can be the elements located on the right side of the periodic table, which are mainly liquids, gases or solids. The electron content in the outermost shell is 4, 5, 6, and 7. Non-Metal is any substance that generates negative ions by accepting electrons. Let’s understand this with the Non-Metal definition and Non-Metal examples. The Non-Metal is only a few in the periodic table, which is presented on the right side. Along with that, it didn’t have a high melting and boiling point. This means it cannot be beaten into sheets of firm wires and even doesn’t conduct electricity or heat like metal does, except in the case of hydrogen, which is a Non-Metal but under high pressure it acts as an alkali metal. In simple words, non-metal is an element that does not show any properties or attributes of metal. They generally have 4 to 7 electrons in their outer shell. A Non-Metal is an element that, by gaining electrons, makes negative ions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
